Tamil Nadu honours WHO Chief Scientist Soumya Swaminathan with special award!
Galatta | Aug 16, 2020, 01:53 pm

India is the third most affected nation by the COVID-19 Novel Corona Virus Pandemic. In our country, Tamil Nadu is the second most affected state and the number of people infected by the Virus is still going up. However, the state has been managing the spread of the Pandemic effectively and the recovery rate has been very high! As of now, Tamil Nadu holds the record for most number of samples tested, across India. Heavy testing and strict isolation and lockdown implementation along with door-to-door testing, has helped bring the Virus spread under control, mainly in the capital city of Chennai. It is well known that the World Health Organisation's (WHO) Chief Scientist Soumya Swaminathan, hails from Tamil Nadu!
Earlier, Chief Scientist Soumya Swaminathan had been very instrumental in guiding the TN Government in her advisory role, to combat the COVID-19 Pandemic, in the state! In recognition of all her endeavours, Soumya Swaminathan was honoured with the Tamil Nadu Chief Minister's Special Award, at the Independence Day celebrations at Fort George, in Chennai. The Chief Minister Edappadi K Palaniswami himself presented her with the award, at the 74th Independence Day celebrations. Soumya Swaminathan had been regularly advising the State health authorities on different measures to follow, to fight the spread of the Pandemic and bring it under control!
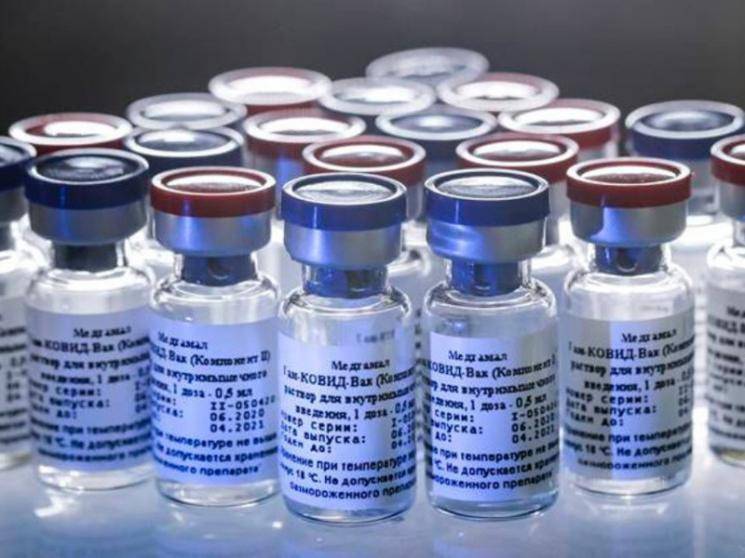
Later, while addressing the Press & Media, the WHO representative thanked the TN Government for the recognition and also urged all the people to continue following the safety precautions mandated by the health authorities. Talking about vaccines, Soumya Swaminathan said that it would take atleast a year and a half for a proper vaccine to be ready, as usually they take about five to ten years but due to the Pandemic, the researchers are rushing the process. She also mentioned that there are about 8 vaccines being developed in India, but that all of them were still in the first stage of clinical testing only and that they would still take time to be ready for production.

.jpg)